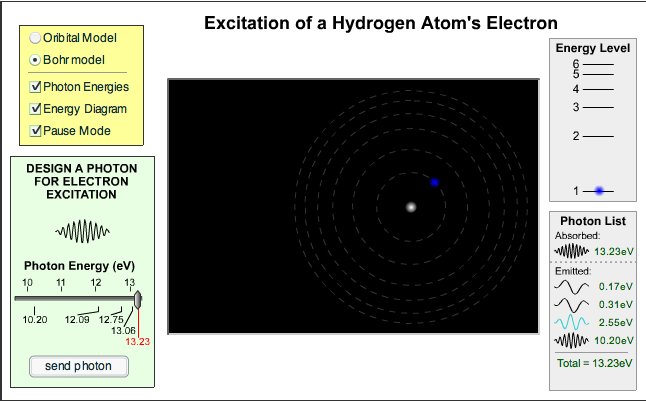
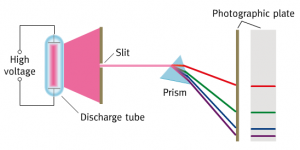
We looked at line spectra with a spectrometer before the holidays and this week we considered how these different colours of light are produced.
Here is a website that lets you choose the energy of a photon and see whether or not it causes a change in the energy of an electron inside the hydrogen atom.
You can read more about line spectra and where they come from here and here.
The visible line spectrum of the Hydrogen atom is explained in the following short film. Click on the image below to start the clip.
I’ve attached a pdf file with further notes on line spectra and the absorption/emission of photons.