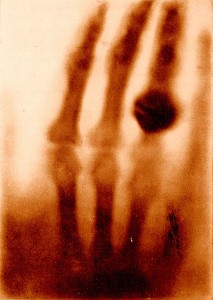
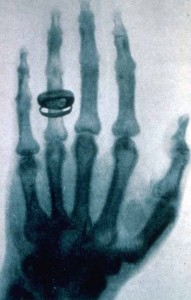
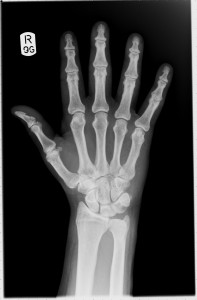
This week, we’ve looked at calculating radiation doses. The absorbed dose D, measured in Grays (Gy), takes into account the energy E absorbed and the mass m of the absorbing tissue.
The higher the energy, the greater the absorbed dose. If you are wondering why the absorbing mass is important, consider the different masses of tissue involved in a dental x-ray and a chest x-ray….
We also learned about equivalent dose in Sieverts (Sv). The equivalent dose H gives an indication of the potential for biological harm by considering the absorbed dose D and a weighting factor .
Different types of radiation have different weighting factors, e.g.
| type of radiation | weighting factor |
|---|---|
| gamma | 1 |
| x-ray | 1 |
| beta | 1 |
| alpha | 20 |
The more damaging forms of radiation have a larger weighting factor.
Absorbed dose and equivalent dose are usually expressed in smaller units; μGy, mGy, μSv, mSv.
In the UK, the population receives an average equivalent dose of 2.2mSv per year due to background radiation produced by cosmic rays, radon gas and materials dug up from the Earth’s crust, such as rocks and soil. In addition to this exposure to background radiation, the Government has set a further equivalent dose of 1mSv per year for members of the public. This limit can be increased to 20mSv for people who work in the nuclear industry, certain medical occupations (such as radiographers) and airline pilots – all of whom will exceed the public limit in the course of their job.
This occupational increase for some individuals can be justified on the grounds that workers are not as vulnerable to the effects of radiation exposure since they are neither children (high rate of cell division so more chance of dna damage being copied) or elderly (reduced ability to repair damage). In many cases, these workers will also be screened on a regular basis by occupational health staff at their place of work.
Here is a poster from the excellent xkcd site that explores examples of the different levels of equivalent dose.
Click on the picture for a larger version.
source: XKCD
Notice that the scale changes as you move through the poster from blue to green to red.
The dosimetry topic is comprehensively covered at BBC Bitesize.