X-rays are a form of electromagnetic radiation. They have a much higher frequency than visible light or ultraviolet. The diagram below, taken from Wikipedia, shows where x-rays sit in the electromagnetic spectrum.

image by Materialscientist
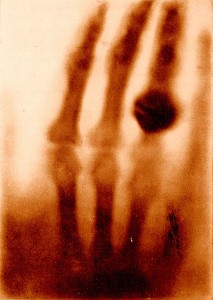
Wilhelm Röntgen discovered x-rays and the image below is the first x-ray image ever taken. It shows Mrs. Röntgen’s hand and wedding ring. The x-ray source used by Röntgen was quite weak, so his wife had to hold her hand still for about 15 minutes to expose the film. Can you imagine waiting that long nowadays?
This was the first time anyone had seen inside a human body without cutting it open. Poor Mrs. Röntgen was so alarmed by the sight of the image made by her husband that she cried out “I have seen my death!” Or, since she was in Germany, it might have been
“Ich habe meinen Tod gesehen!“
that she actually said.
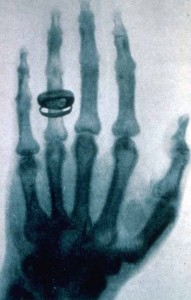
Röntgen continued to work on x-rays until he was able to produce better images. The x-ray below was taken about a year after the first x-ray and you can see the improvements in quality.
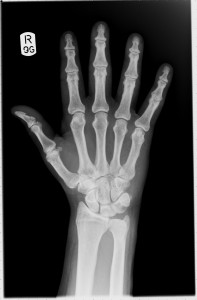
Notice that these early x-rays are the opposite of what we would expect to see today. They show dark bones on a lighter background while we are used to seeing white bones on a dark background, such as the x-ray shown below. The difference is due to the processing the film has received after being exposed to x-rays.
In hospitals, x-rays expose a film which is then developed and viewed with bright light. X-rays are able to travel through soft body tissue and the film behind receives a large exposure.
The x-rays darken the film.
More dense structures such as bone, metal fillings in teeth, artificial hip/knee joints, etc. block the path of x-rays and prevent them from reaching the film. Unexposed regions of the film remain light in colour.
Röntgen’s x-ray films would have involved additional processing steps. The exposed films were developed and used to create a positive. In creating a positive, light areas become dark and dark areas become light. So the light and dark areas in Röntgen’s x-rays are the opposite of what we see today. Our modern method makes it easier to detect issues in the bones as they are the lighter areas.
Röntgen was awarded the first ever Nobel Prize for Physics in 1901 for his pioneering work in this field of physics.
X-rays are a very high energy form of electromagnetic radiation. This means they have the potential to harm living cells. Medical staff only take x-rays of a patient when it is necessary to give a correct diagnosis but that wasn’t always the case.
I have attached a recording of a short BBC radio programme about the first x-ray and what people in the Victorian era thought of these new images. Click on the player at the end of this post.